
If there are no lone electron pairs on the central atom, the electron pair and molecular geometries are the same.Ĭlick here to see the various molecular geometries.Ĭhoose the correct molecular geometries for the following molecules or ions below. 
Step 4: The molecular geometry describes the position only of atomic nuclei (not lone electron pairs) of a molecule (or ion). * Lone electron pairs are represented by a line without an atom attached. We review their content and use your feedback to keep the quality high. Who are the experts Experts are tested by Chegg as specialists in their subject area. The table below shows the electron pair geometries for the structures we've been looking at: The electron pair geometry for ClF 3 is and its molecular geometry is. The regions of electron density will arrange themselves around the central atom so that they are as far apart from each other as possible. 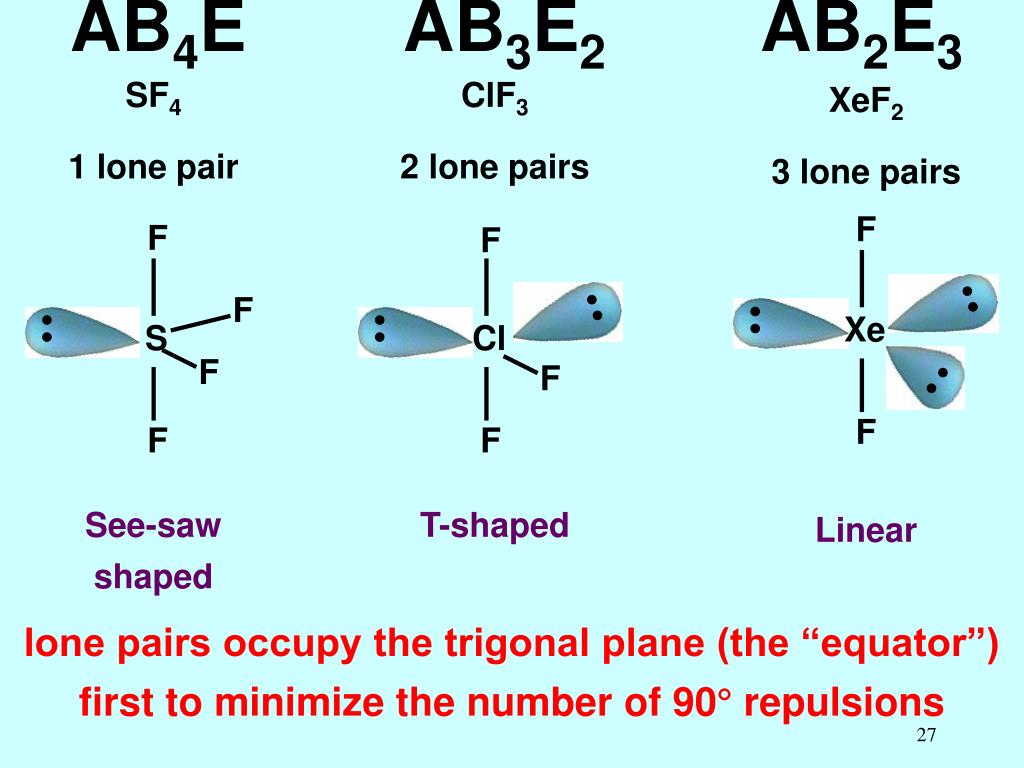
Use this number to determine the electron pair geometry.Įach bond (whether it be a single, double or triple bond) and each lone electron pair is a region of electron density around the central atom. 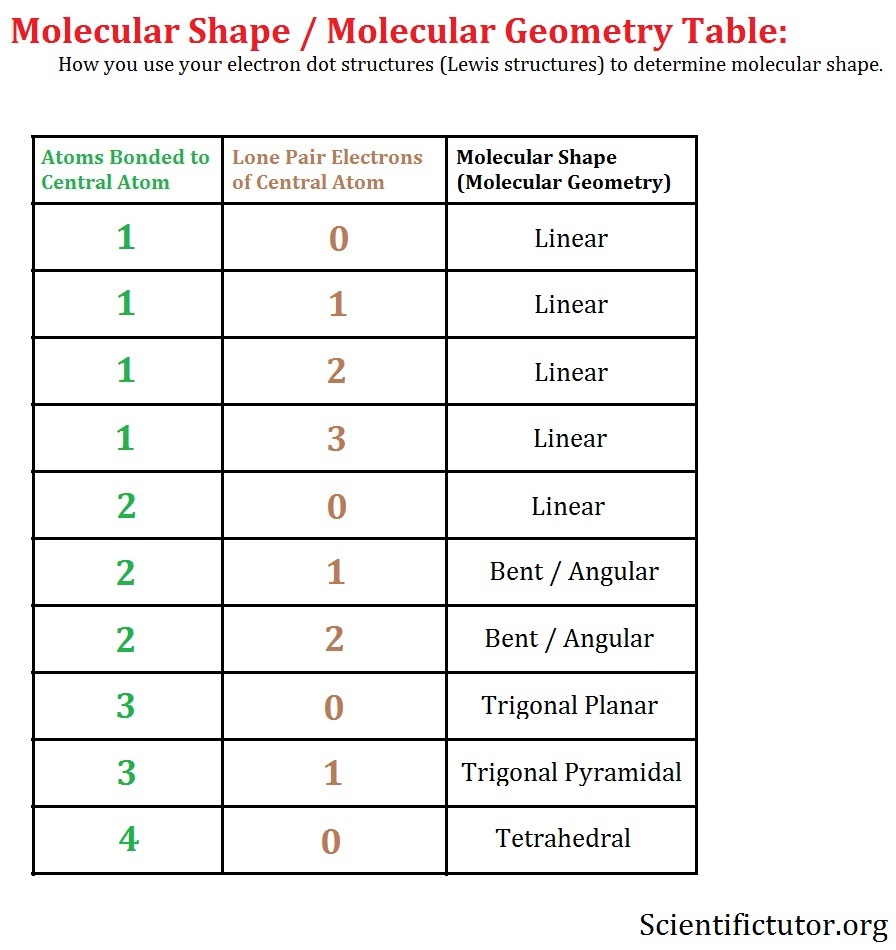
Step 3: Add these two numbers together to get the regions of electron density around the central atom. The valency of chlorine (Cl) is dependent on the other atoms to which it is bonded. As it is known that chlorine (Cl) can have more than one valency. Thus, ClF3 is T-shaped, having the axial FClF compressed from the angle of 180°, which is a formal sp3d hybridization. Make sure you understand why they are correct. We can describe the molecular geometry in terms of atoms, but not electrons. The correct answers have been entered for you. How many lone electron pairs are on the central atom in each of the following Lewis structures? Step 1: Count the number of lone electron pairs on the central atom. Molecular geometry can be predicted using VSEPR by following a series of steps: In other words, the electrons will try to be as far apart as possible while still bonded to the central atom. The theory says that repulsion among the pairs of electrons on a central atom (whether bonding or non-bonding electron pairs) will control the geometry of the molecule. The valence shell electron-pair repulsion theory (abbreviated VSEPR) is commonly used to predict molecular geometry. Lewis structures are very useful in predicting the geometry of a molecule or ion. That leaves one equatorial, and 2 axial, sites where the Cl-F bonds can go, which leaves you with a T-shaped geometry, rather than a trigonal planar structure.Many of the physical and chemical properties of a molecule or ion are determined by its three-dimensional shape (or molecular geometry). Therefore, the interactions are lowest in the equatorial environment, so this is where the lone pairs go. In an equatorial environment however, only 2 bonds/lp's are at a 90 degree angle (the axial ones), with 2 at 120 degree angles. In an axial environment, there are 3 bonds/lp's at a 90 degree angle (the equatorial ones), with 1 at 180 degrees (the other axial one). In a basic trigonal bipyramidal structure, you've got 2 environments, an axial and an equatorial one: Therefore, to minimise the energy of the molecule, the lone pairs need putting in whichever position minimises their interactions. In a lone pair (lp), the average position of the electrons is closer to the central atom than the electrons involved in a covalent (in this case Cl-F) bond, so they have a slightly greater repulsion to other lone pairs (or bonds), than a bond does.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
